
The treatment of central sleep apnea (CSA) can be challenging. An American Academy of Sleep Medicine (AASM) task force recently performed an extensive review of published data and developed guidelines on CSA management. This article provides a summary of those recommendations, including the challenging decision of whether adaptive servo-ventilation (ASV) is a safe option in these patients.
Background
Per the International Classification of Sleep Disorders (ICSD), central sleep apnea is defined by five or more central apneas and/or hypopneas per hour, with > 50% of events being central in nature and associated symptoms such as sleepiness or insomnia.1 While the ICSD categorizes CSA into six diagnostic categories (primary CSA, CSA with Cheyne-Stokes respiration [CSR], CSA due to a medical disorder without CSR, CSA due to medication or substance, treatment-emergent CSA, and CSA due to high altitude),the task force opted to group studies that evaluated similar treatments across the different CSA subtypes.1–2 They did this for the following reasons: there is a limited number of studies available; many studies do not differentiate CSA subtypes; and CSA, while triggered by different pathways, shares a common final pathway that involves post-hyperventilation hypocapnia (equifinality).2

Overview of new guidelines
Task force members reviewed relevant research published before February 2025 using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) method.3 They looked for a clinically meaningful effect of the intervention on what was determined to be critical outcomes: disease severity (as measured by apnea-hypopnea index [AHI], central apnea index [CAI], central apnea hypopnea index [CAHI], and oxygen desaturation index [ODI]), symptoms, function, and clinical outcomes (eg, hospitalizations and mortality).2 Based on the quality of evidence available, all the recommendations the task force made on the various interventions were marked as conditional—suggesting that providers offer these treatments to patients with an individualized approach, based on clinical context.2
The guidelines emphasize that CSA treatment should focus on reducing respiratory events and improving symptoms, as well as addressing its underlying etiology.2 Potential options for treating CSA include PAP therapy, supplemental oxygen, acetazolamide, and transvenous phrenic nerve stimulation (TPNS).2 Rather than proposing a specific algorithm, the guidelines suggest that if one intervention does not work then it is reasonable to try another intervention.2 (See Table 1 for the specific GRADE recommendations for each intervention by the task force.)
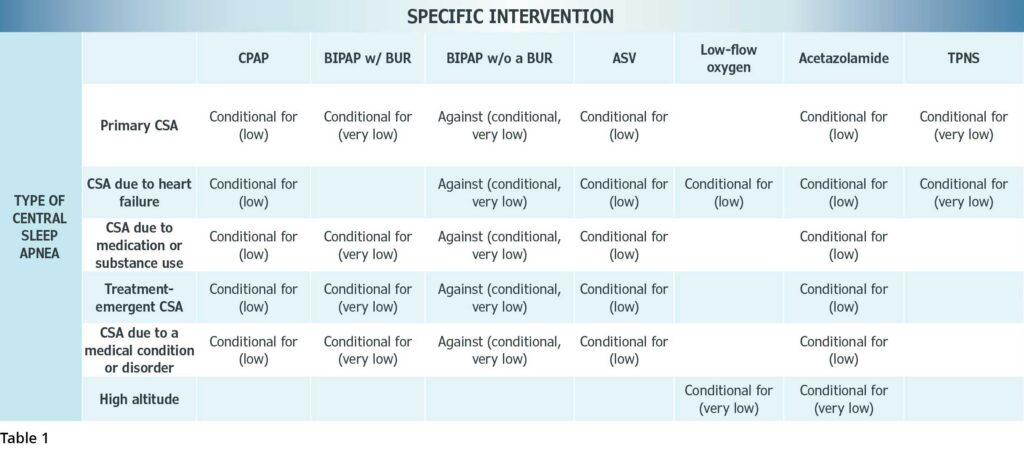
CPAP is a reasonable initial treatment for most types of CSA since it has been shown to reduce AHI and is readily available.2 Use of BiPAP without a backup rate (BUR) is not advised, however, since it has the potential to induce central apneas.2 On the other hand, a conditional recommendation for BiPAP with a BUR was given for treatment of CSA (except when secondary to heart failure and high altitude, due to lack of evidence in these subgroups).2 This was based on their analysis of six randomized controlled trials (RCTs) that found BiPAP with a BUR improved sleepiness, AHI, CAI, CAHI, left ventricular ejection fraction (LVEF), and heart rate.3 The guidelines also tackled a controversial management challenge in sleep medicine—the use of adaptive servo-ventilation (ASV)—ultimately giving it a conditional recommendation for management of most types of CSA.2
Low-flow supplemental oxygen received a conditional recommendation for the treatment of CSA due to heart failure and high altitude.2 In CSA due to heart failure, supplemental oxygen has been shown to improve AHI, CAI, sleep quality, and hospitalizations.2 In CSA related to high altitude, supplemental oxygen reduces the ODI.2 Practically, however, insurance coverage may be a barrier in using supplemental oxygen to treat CSA.2
Acetazolamide was the only intervention that received a conditional recommendation for all six classifications of CSA, with doses ranging from 250 mg to 1,000 mg shown to reduce the AHI.2 The task force determined that the potential side effects of acetazolamide were mild (paresthesia, change in taste, kidney stones, etc) when compared with potential benefits.2 However, there were concerns on the changes in acid-base status, ventilation, and electrolytes, as well as questions on the dosage and duration of therapy.2–3 Most studies reported only short-term outcomes.2 The task force suggests that if using acetazolamide for treatment, there should be close monitoring of symptoms and side effects as well as repeat testing.2
Transvenous phrenic nerve stimulation (TPNS) was given a conditional recommendation for management of primary CSA and CSA secondary to heart failure, driven primarily by a single RCT showing that this therapy improved sleepiness, reduced CAI by 80%, increased 6-minute walk test distance, and decreased arousal index.3 Of note, TPNS has not been shown to reduce mortality or improve LVEF.3 Feasibility of TPNS implantation can be challenging, however, given limited expertise, availability, the need for coordination with other specialties, and overall cost (> $50,000).3–
Compared with the 2016 guidelines, some of the major changes were the addition of TPNS and comments on ASV.2,4 Other changes were related to the task force’s determination that based on available data they could not comment on the use of hypnotics in the treatment of CSA as well as BiPAP with a BUR for CSA due to heart failure; previous guidelines gave an “option” recommendation.2,4 Similarly, there were no longer any comments regarding treatment of CSA in patients with end-stage renal disease.
Conditional recommendation for ASV
The decision on the use of ASV to treat CSA in heart failure with reduced ejection fraction (HFrEF) has been a major challenge in the sleep medicine world for over a decade. The 2016 guidelines recommended against the use of ASV in symptomatic heart failure patients with LVEF of ≤ 45%.4 This stemmed from the Treatment of Predominant Central Sleep Apnoea by Adaptive Servo Ventilation in Patients With Heart Failure (SERVE-HF) trial, which found a 34% increased risk of cardiovascular death in CSA patients with LVEF of ≤ 45% who used ASV.5 However, publication of the 2024 Effect of Adaptive Servo Ventilation (ASV) on Survival and Hospital Admissions in Heart Failure (ADVENT-HF) trial caused many to consider this recommendation. The ADVENT-HF data found no increase in mortality on three- to six-year follow-up when using an ASV from a different manufacturer with a peak flow target to treat both obstructive and central sleep apnea in patients with HFrEF.6
After SERVE-HF, BiPAP with a BUR became the frequent go-to in treating CSA with HFrEF. However, the new guidelines do not provide any recommendation on BiPAP with a BUR for CSA treatment in patients with heart failure. Instead, the guidelines suggest ASV to treat heart failure patients with CSA without a specific LVEF cutoff.2 This may pose some problems with some payors who, in our experience, will cover ASV only when BiPAP with a BUR has been tried and fails to control CSA.
The task force members’ recommendations on ASV were based on the analysis of 12 RCTs.3 Their analysis found that ASV resulted in > 70% reduction in AHI, CAI, CAHI, and/or ODI.3 They did find an increased signal of hospitalization in patients using ASV but deemed this not to be clinically meaningful.3 When pooling the data from four trials (including SERVE-HF and ADVENT-HF), the task force found that the use of ASV in HFrEF patients was not associated with increased mortality.3 In weighing the risks and benefits of ASV therapy, they determined that since the ASV device used in the SERVE-HF trial that showed increased mortality is no longer manufactured, the benefits of ASV as a class outweigh the risks; thus, ASV received a conditional recommendation for use with low certainty of evidence.2
The conditional recommendation on ASV was presented with some caution.2 It was acknowledged that ASV devices are made by different manufacturers and that each has a different algorithm.2 Since the task force looked at ASV as one general class rather than by brand, it could not recommend a specific ASV brand.2 For these reasons, the task force emphasized that the use of ASV involves shared decision-making with the patient, close follow-up, and utilization by experienced centers.2
Future directions
One clear theme that emerged from the newly released AASM guidelines is that more data is needed to be able to provide strong recommendations. Some of the limitations in the current data are that studies sometimes lumped different CSA disorders together when evaluating an intervention and that central hypopneas were often not adequately measured or identified in the studies.3 Other considerations that affect treatment recommendations are that comorbid OSA can make it difficult to recognize and treat CSA and that a class of device might work differently based on the manufacturer.3
There is need for future research concerning targeted therapies based on etiology or pathophysiology, analysis of whether CSR is a nonharmful compensatory mechanism, understanding when and how CPAP improves CSA, looking at the use of multimodal therapy, comparing different treatment modalities, and treatment strategies that improve mortality.3
As we await new research, the task force has provided a helpful step forward addressing interventions that sleep medicine providers can consider for patients with CSA.
This article was originally published in the Spring 2026 issue of CHEST Physician.
References
1. The AASM international classification of sleep disorders. American Academy of Sleep Medicine. 3rd ed, text rev. Darien, IL: American Academy of Sleep Medicine; 2023.
2. Badr MS, Khayat RN, Allam JS, Hyer S, Mustafa RA, Naughton MT, Patil S, Pien GW, Randerath W, Won C. Treatment of central sleep apnea in adults: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2025;21(12):2181-2191. doi:10.5664/jcsm.11858
3. Badr MS, Khayat RN, Allam JS, Hyer S, Mustafa RA, Naughton MT, Patil S, Pien GW, Randerath W, Won C. Treatment of central sleep apnea in adults: an American Academy of Sleep Medicine systematic review, meta-analysis, and GRADE assessment. J Clin Sleep Med. 2025;21(12):2213-2236. doi:10.5664/jcsm.11860
4. Aurora RN, Bista SR, Casey KR, et al. Updated adaptive servo-ventilation recommendations for the 2012 AASM guideline: the treatment of central sleep apnea syndromes in adults: practice parameters with an evidence-based literature review and meta-analyses. J Clin Sleep Med. 2016;12(5):757-61. doi:10.5664/jcsm.5812
5. Cowie MR, Woehrle H, Wegscheider K, et al. Adaptive servo-ventilation for central sleep apnea in systolic heart failure. N Engl J Med. 2015;373(12):1095-1105. doi:10.1056/NEJMoa1506459
6. Bradley TD, Logan AG, Lorenzi Filho G, et al. Adaptive servo-ventilation for sleep-disordered breathing in patients with heart failure with reduced ejection fraction (ADVENT-HF): a multicentre, multinational, parallel-group, open-label, phase 3 randomised controlled trial. Lancet Respir Med. 2024;12(2):153-166. doi:10.1016/S2213-2600(23)00374-0
