
Epidemiology and global burden of post-TB lung disease
As of 2020, there were an estimated 155 million TB survivors worldwide. TB accounted for approximately 122 million disability-adjusted life years (DALYs) globally. Nearly half, about 58 million DALYs, were attributable to post-TB morbidity rather than active disease, causing profound clinical and socioeconomic impacts, particularly in low- and middle-income countries.1–2 Among the patients diagnosed with TB in the United States, a loss of 48% of quality-adjusted life expectancy was estimated to result from post-TB sequelae.3
Variation in the definition of post-TB lung disease (PTLD) across research studies limits the interpretation and synthesis of evidence. A recently proposed case definition includes three components: history of previous pulmonary or pleural TB, absence of active TB, and abnormalities in at least two of three clinical domains of lung function, respiratory symptoms, and chest imaging, in part attributed to previous TB.4 TB survivors face a threefold to sixfold higher mortality rate, and most deaths occur within the first year following treatment completion, with cardiovascular disease and cancer being the most common causes.2,5–6

Pathophysiology, clinical patterns, and long-term complications
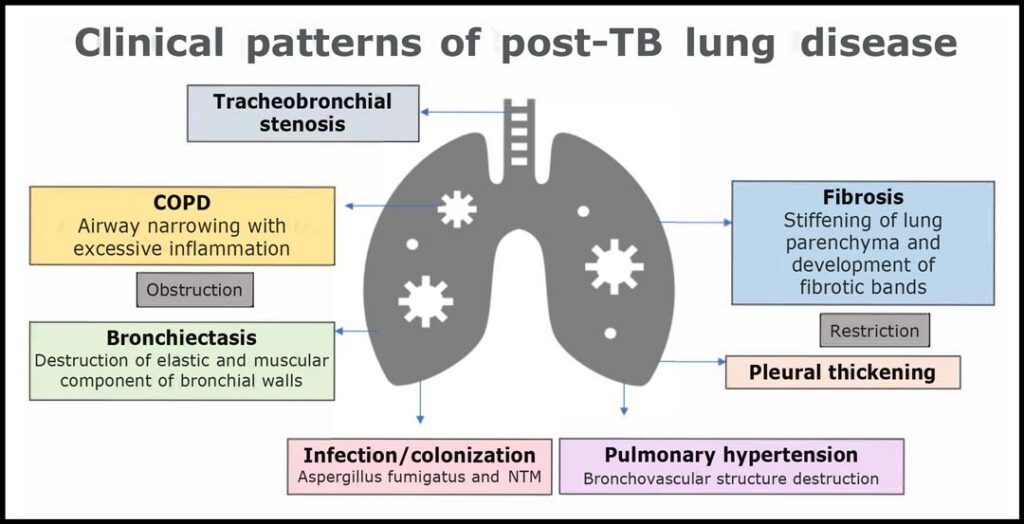
PTLD is affected by several factors: a) Disease site, infection burden, resistance pattern, and rate of clearance, b) Host immune response, causing excessive remodeling and fibrosis, and comorbidities, c) Environmental exposures (pollutants), tobacco use, and recurrent bacterial and viral infections.6–7 TB can thus affect multiple pulmonary compartments, including the lung parenchyma, large and small airways, vasculature, and pleura.5
Bronchiectasis is one of the most common structural sequelae of TB, frequently involving the upper lobes and often creating a vicious cycle of inflammation and infection, along with increasing risk for complications like chronic pulmonary aspergillosis in the destroyed lung tissue.8 TB-associated COPD can affect up to half of patients with PTLD, with small airways disease causing gas to be trapped and limiting airflow. It disproportionately affects younger patients. Delay in anti-TB treatment initiation has also been associated with increased risk of COPD development, suggesting that some TB-associated COPD cases can be preventable with early TB diagnosis and management.8–10
Additionally, pleural fibrosis and fibrothorax can lead to restrictive physiology, which affects up to 25% of survivors.11 Survivors of TB are 2.17 times more likely to develop lung cancer, and TB is associated with increased mortality.12 Pulmonary hypertension was suspected in two-thirds of patients based on radiological findings, though this requires further validation in future studies.11 Up to 40% of patients can have endobronchial TB, which can cause airway stenosis.1 Survivors of TB also face increased risk of recurrent TB disease, whether from relapse or reinfection.5

Diagnosis and assessment of post-TB lung damage
Diverse clinical patterns and disease trajectories, along with heterogeneous definitions of PTLD, require an individualized approach to patients, starting with a detailed symptom assessment and systematic evaluation.5 Up to 40% of survivors are symptomatic, and dyspnea impacts their quality of life.13 Functional and radiological evaluation remains critical for diagnosis.6
PTLD can cause obstructive, restrictive, or mixed patterns of functional abnormalities on spirometry, and abnormal spirometry is seen in almost 60% of post-TB patients, with 10% to 15% of survivors demonstrating severe impairment in pulmonary function.1,5
Abnormal lung imaging was evident in the majority of patients with PTLD. High-resolution CT scan helps identify specific patterns of structural damage, including bronchiectasis, cavitation, fibrosis, and emphysema.14–15 Multidrug-resistant TB-associated PTLD has a higher prevalence of impaired lung function, radiological sequelae, and mortality.6
Management and treatment strategies
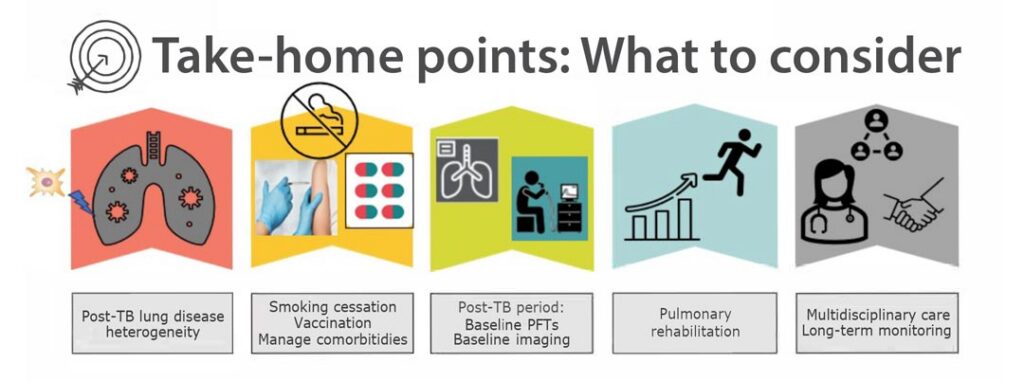
Baseline and periodic assessment of symptom severity using approved dyspnea scales, spirometry, and ambulatory oximetry, as well as monitoring for TB recurrence, remains the basis of post-TB care.1 Potential interventions include adequate nutritional support, chest physiotherapy, vaccinations, pulmonary rehabilitation, and smoking cessation counseling.
Pulmonary rehabilitation is recommended to improve lung function and exercise capacity, as well as address general deconditioning and muscle loss. It remains the cornerstone therapy for chronic respiratory diseases, including PTLD.1,5,16 Bronchodilator therapy appears comparably efficacious in TB-associated obstructive lung physiology, but the use of inhaled corticosteroids is discouraged due to increased risk of infection.1,8–9 Since smoking exacerbates lung destruction, smoking cessation counseling is extremely important. Vaccinations against pneumococcus and influenza are routinely recommended.1,5,17
Secondary complications of PTLD may require targeted interventions. Studies of patients with all-cause noncystic fibrosis bronchiectasis in high TB burden settings suggest a high burden of gram-negative lung infections, and there is an increased risk of aspergilloma and invasive pulmonary aspergillosis in patients with residual cavitary lung disease. Surgical intervention may be considered in selected cases with severe tracheobronchial stenosis, structural lung disease, or complications like hemoptysis with severe bronchiectasis.1,13
Future directions in PTLD care
The recognition of PTLD as a major contributor to global chronic lung disease burden necessitates integration of post-TB care into national TB control programs, with emphasis on prevention, early disease detection, and comprehensive long-term management strategies. Multidisciplinary care is essential to addressing respiratory, psychological, and socioeconomic consequences.6,17 Creating standardized diagnostic criteria, screening protocols, and therapeutic management pathways for PTLD remain urgent research priorities.1,5–6
References
1. Seo W, Kim HW, Kim JS, et al. Long term management of people with post-tuberculosis lung disease. Korean J Intern Med. 2024;39(1):7-24. doi:10.3904/kjim.2023.395
2. Wang J, Yuan B, Fang Y, et al. Post-tuberculosis morbidities and their associated mortality: moving from challenges to solutions. Eur Respir Rev. 2025;34(178):250148. doi:10.1183/16000617.0148-2025
3. Menzies NA, Marks SM, Hsieh YL, et al. Contribution of posttuberculosis sequelae to life-years lost from tuberculosis disease in the united states, 2015-2019. Am J Respir Crit Care Med. 2025;211(6):1059-1068. doi:10.1164/rccm.202411-2213OC
4. Bisson GP, Allwood B, Byrne A, et al. Post-tuberculosis lung disease: a case definition for use in research studies. Lancet Infect Dis. 2025. doi:10.1016/S1473-3099(25)00548-1
5. Meghji J, Auld SC, Bisson GP, et al. Post-tuberculosis lung disease: towards prevention, diagnosis, and care. Lancet Respir Med. 2025;13(5):460-472. doi:10.1016/S2213-2600(24)00429-6
6. Fumagalli G, Mencarini J, Sini I, et al. Post-tuberculosis lung disease: a guide for clinicians. Infection. Sept. 25, 2025. doi:10.1007/s15010-025-02645-2
7. Auld SC, Barczak AK, Bishai W, et al. Pathogenesis of post-tuberculosis lung disease: defining knowledge gaps and research priorities at the second international post-tuberculosis symposium. Am J Respir Crit Care Med. 2024;210(8):979-993. doi:10.1164/rccm.202402-0374SO
8. Sehgal IS, Dhooria S, Muthu V, et al. Burden, clinical features, and outcomes of post-tuberculosis chronic obstructive lung diseases. Curr Opin Pulm Med. 2024;30(2):156-166. doi:10.1097/MCP.0000000000001026
9. Gai X, Allwood B, Sun Y. Post-tuberculosis lung disease and chronic obstructive pulmonary disease. Chin Med J (Engl). 2023;136(16):1923-1928. doi:10.1097/CM9.0000000000002705
10. Lee CH, Lee MC, Lin HH, et al. Pulmonary tuberculosis and delay in anti-tuberculous treatment are important risk factors for chronic obstructive pulmonary disease. PLoS One. 2012;7(5):e37978. doi:10.1371/journal.pone.0037978
11. Byrne A, Al-Hindawi Y, Plit M, et al. The prevalence and pattern of post tuberculosis lung disease including pulmonary hypertension from an Australian TB service; a single-centre, retrospective cohort study. BMC Pulm Med. 2025;25(1):84. doi:10.1186/s12890-025-03549-5
12. Abdeahad H, Salehi M, Yaghoubi A, et al. Previous pulmonary tuberculosis enhances the risk of lung cancer: systematic reviews and meta-analysis. Infect Dis (Lond). 2022;54(4):255-268. doi:10.1080/23744235.2022.2045630
13. Taylor J, Bastos ML, Lachapelle-Chisholm S, et al. Residual respiratory disability after successful treatment of pulmonary tuberculosis: a systematic review and meta-analysis. EClinicalMedicine. 2023;59:101979. doi:10.1016/j.eclinm.2023.101979
14. Meghji J, Simpson H, Squire SB, et al. A systematic review of the prevalence and pattern of imaging defined post-TB lung disease. PLoS One. 2016;11(8):e0161176. doi:10.1371/journal.pone.0161176
15. Perumal R, Pillay S, Bagratee N, et al. Anatomical and functional characteristics of symptomatic post-TB lung disease. Int J Tuberc Lung Dis. 2025;29(10):434-440. doi:10.5588/ijtld.25.0137
16. Migliori GB, Marx FM, Ambrosino N, et al. Clinical standards for the assessment, management and rehabilitation of post-TB lung disease. Int J Tuberc Lung Dis. 2021;25(10):797-813. doi:10.5588/ijtld.21.0425
17. Allwood BW, Byrne A, Meghji J, et al. Post-tuberculosis lung disease: clinical review of an under-recognised global challenge. Respiration. 2021;100(8):751-763. doi:10.1159/000517371
