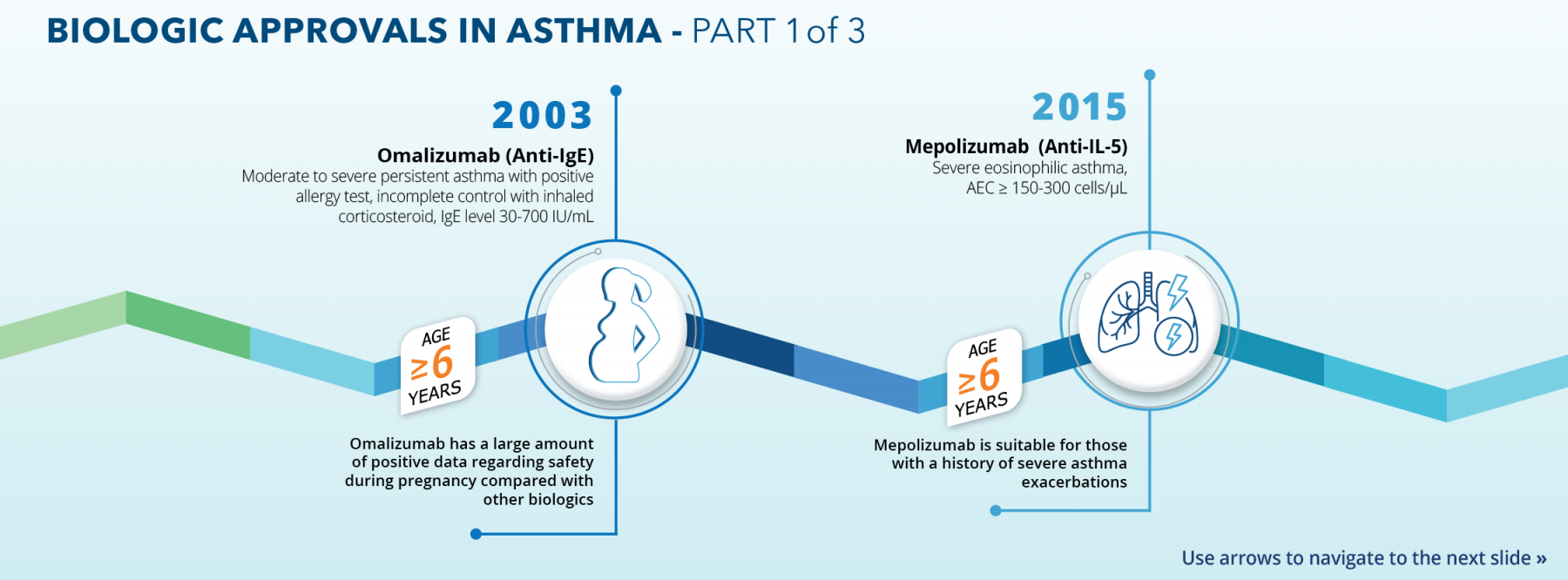
Asthma affects approximately 350 million people of all ages worldwide, with approximately 5% to 10% of those experiencing severe disease.1–2 Type 2 mediated disease accounts for the vast majority—up to 80%—of cases.2–3 Biologic therapies have had a profound impact on disease management in patients with moderate to severe asthma. These agents have been shown to decrease exacerbations and the reliance on oral steroids, improve lung function and quality of life in a subset of patients, and even reduce the need for inhaled corticosteroid maintenance therapy.4–8

Overall, this has led to a paradigm shift in asthma management goals from control to remission across patients with varying levels of disease severity. Achieving remission has become an aspirational goal for asthma management. However, only about one-third of patients receiving biologics can achieve remission.9–10 Thus, there is still a need to improve the approach for patients with severe asthma.
Despite the lack of comparative effectiveness trials across currently available biologic therapies, a new CHEST guideline on the use of biologics for severe disease was created to assist with therapeutic decision-making based on individual patient characteristics.11 As research continues to demonstrate enduring efficacy for these agents, clinicians should be encouraged to make greater use of biologic therapies for their patient populations, especially for those with more severe disease or with comorbid conditions such as chronic rhinosinusitis with nasal polyps.

More recently, radiographic findings have been found to be important for identifying phenotypes in asthma. For example, mucus plugging seen on chest CT scans has been linked to severe asthma and acute exacerbations, sometimes resulting in death.12 Patients who have evidence of mucus plugging have been shown to have greater type 2 inflammation and more frequent severe exacerbations.13–14
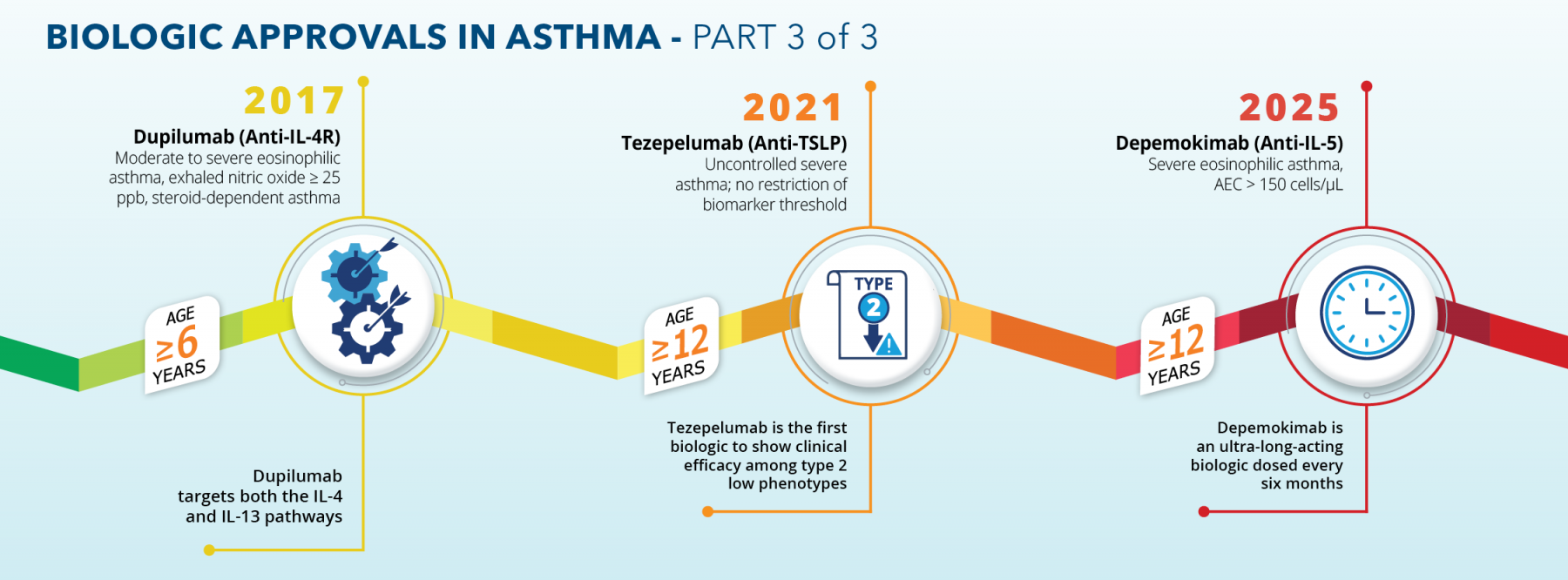
Novel research, such as the VESTIGE trial, has shown that biologic therapies may have a greater response for these patients.15 Such research demonstrates how the addition of radiographic assessments can further advance patient care.16 Looking ahead, ultra-long-acting biologics are poised to change the treatment paradigm in severe asthma and may provide sustained control of type 2 inflammation with less frequent dosing.17–18 (see “Adding to the toolkit: Depemokimab”)

Adding to the toolkit: Depemokimab
Depemokimab, an interleukin-5 (IL-5) targeted agent, was recently approved (December 2025) for use by the US Food and Drug Administration and is the first ultra-long-acting biologic approved for severe asthma.
In the phase 3A, randomized, placebo-controlled replicate SWIFT-1 and SWIFT-2 trials, the pooled annualized rate of exacerbations was 0.51 (95% CI, 0.43-0.60) compared with 1.11 (95% CI, 0.92-1.33) for placebo. In addition, the every-six-month dosing is expected to result in improved patient quality of life.17–18
This article was originally published in the Spring 2026 issue of CHEST Physician.
References
1. Shah PA, Brightling C. Biologics for severe asthma—which, when and why? Respirology. 2023;28(8):709-721. doi:10.1111/resp.14520
2. Papi A, Brightling C, Pedersen SE, Reddel HK. Asthma. Lancet. 2018; 391(10122):783–800. doi:10.1016/S0140-6736(17)33311-1
3. Rogers L, Jesenak M, Bjermer L, et al. Biologics in severe asthma: a pragmatic approach for choosing the right treatment for the right patient. Respir Med. 2023;218:107414. doi:10.1016/j.rmed.2023.107414
4. Korn S, Bourdin A, Chupp G, et al. Integrated safety and efficacy among patients receiving benralizumab for up to 5 years. J Allergy Clin Immunol Pract. 2021;9(12):4381-4392.e4. doi:10.1016/j.jaip.2021.07.058
5. Jackson DJ, Heaney LG, Humbert M, et al. Reduction of daily maintenance inhaled corticosteroids in patients with severe eosinophilic asthma treated with benralizumab (SHAMAL): a randomised, multicentre, open-label phase 4 study. Lancet. 2024;403(10423):271-281. doi:10.1016/S0140-6736(23)02284-5
6. Thomas D, McDonald VM, Stevens S, et al. Biologics (mepolizumab and omalizumab) induced remission in severe asthma patients. Allergy. 2024;79(2):384-392. doi:10.1111/all.15867
7. Hansen S, Søndergaard MB, von Bülow A, et al. Clinical response and remission in patients with severe asthma treated with biologic therapies. Chest. 2024;165(2):253-266. doi:10.1016/j.chest.2023.10.046
8. Castro M, Corren J, Pavord ID, et al. Dupilumab efficacy and safety in moderate-to-severe uncontrolled asthma. N Engl J Med. 2018;378:2486-2496. doi:10.1056/NEJMoa1804092
9. Lugogo NL, Mohan A, Akuthota P, et al. Are we ready for asthma remission as a clinical outcome? Chest. 2023;164(4):831-834. doi:10.1016/j.chest.2023.04.028
10. Blaiss M, Oppenheimer J, Corbett M, et al. Consensus of an American College of Allergy, Asthma, and Immunology, American Academy of Allergy, Asthma, and Immunology, and American Thoracic Society workgroup on definition of clinical remission in asthma on treatment. Ann Allergy Asthma Immunol. 2023;131(6):782-785. doi:10.1016/j.anai.2023.08.609
11. Oberle AJ, Abbas F, Adrish M, et al. Biologic management in severe asthma for adults: an American College of Chest Physicians clinical practice guideline. Chest. Published online September 24, 2025. doi:10.1016/j.chest.2025.08.042
12. Kuyper LM, Paré PD, Hogg JC, et al. Characterization of airway plugging in fatal asthma. Am J Med. 2003;115(1):6-11. doi:10.1016/s0002-9343(03)00241-9
13. Dunican EM, Elicker BM, Gierada DS, et al. Mucus plugs in patients with asthma linked to eosinophilia and airflow obstruction. J Clin Invest. 2018;128(3):997-1009. doi:10.1172/JCI95693
14. Chan R, Duraikannu C, Lipworth B. Clinical associations of mucus plugging in moderate to severe asthma. J Allergy Clin Immunol Pract. 2023;11(1):195-199.e2. doi:10.1016/j.jaip.2022.09.008
15. Castro M, Papi A, Porsbjerg C, et al. Effect of dupilumab on exhaled nitric oxide, mucus plugs, and functional respiratory imaging in patients with type 2 asthma (VESTIGE): a randomised, doubleblind, placebo-controlled, phase 4 trial. Lancet Respir Med. 2025;13:208-220. doi:10.1016/S2213-2600(24)00362-X
16. Götschke J, Walter J, Leuschner G, et al. Mucus plug score predicts clinical and pulmonary function response to biologic therapy in patients with severe asthma. J Allergy Clin Immunol Pract. 2025;13(5):1110-1122. doi:10.1016/j.jaip.2025.01.010
17. Jackson DJ, Wechsler ME, Jackson DJ, et al. Twice-yearly depemokimab in severe asthma with an eosinophilic phenotype. N Engl J Med. 2024;391:2337-2349. doi:10.1056/NEJMoa2406673
18. Press release. Depemokimab applications accepted for review by the US FDA for asthma with type 2 inflammation and for chronic rhinosinusitis with nasal polyps (CRSwNP). Published March 3, 2025
19. Akuthota P, Castro M, Pavord I, et al. Amlitelimab phase 2 clinical trial results in patients with moderate-to-severe asthma. Eur Respir J. 2025;66(suppl 69): OA1180. doi:10.1183/13993003.congress-2025.OA1180
20. Kelsen SG, Agache IO, Soong W, et al. Astegolimab (anti-ST2) efficacy and safety in adults with severe asthma: A randomized clinical trial. J Allergy Clin Immunol. 2021;148(3):790-798. doi:10.1016/j.jaci.2021.03.044
21. A Study to Investigate the Efficacy and Safety of Verekitug (UPB-101) in Adult Participants With Severe Asthma (VALIANT). ClinicalTrials.gov. NCT06196879
22. A Long-Term Safety and Efficacy Study of Verekitug (UPB-101) in Adult Participants With Severe Asthma (VALOUR). ClinicalTrials.gov. NCT06966479
23. Harbour BioMed press release. HBM9378/WIN378, a Long-Acting, Fully Human Anti-TSLP Antibody, Enters Global Phase 2 POLARIS Trial for Asthma. Published July 23, 2025
24. England E, Rees DG, Scott IC, et al. Tozorakimab (MEDI3506): an anti-IL-33 antibody that inhibits IL-33 signalling via ST2 and RAGE/EGFR to reduce inflammation and epithelial dysfunction. Sci Rep. 2023;13:9825. doi:10.1038/s41598-023-36642-y
25. Corren J, Reid F, Moate R, et al. S90 FRONTIER-3: a randomized, phase 2a study to evaluate the efficacy and safety of tozorakimab (an anti-interleukin-33 monoclonal antibody) in early-onset asthma. Thorax. 2024;79(Suppl 2):S90. doi:10.1136/thorax-2024-BTSabstracts.96
26. Deiteren A, Krupka E, Bontinck L, et al. A proof-of-mechanism trial in asthma with lunsekimig, a bispecific NANOBODY molecule. Eur Respir J. 2025;65:2401461. doi:10.1183/13993003.01461-2024
27. Deiteren A, Bontinck L, Conickx G, et al. A first-in-human, single and multiple dose study of lunsekimig, a novel anti-TSLP/anti-IL-13 NANOBODY compound, in healthy volunteers. Clin Transl Sci. 2024;17:e13864. doi:10.1111/cts.13864
28. A Study to Learn More About the Study Medicine PF-07275315 in Healthy Chinese Adult Participants. ClinicalTrials.gov. NCT06675188
29. A Study to Learn About the Study Medicine Called PF-07275315 in People With Moderate-To-Severe Asthma. ClinicalTrials.gov. NCT06977581