Presented with Support from Chiesi
The content of this article and the opinions expressed herein are those of the included experts and not of the supporter. It is not an endorsement by CHEST and does not reflect the views or opinions of CHEST.
SAD in severe asthma that is not well controlled: Understanding the importance of particle size and delivery system
Small airway disease (SAD) has been noted to be particularly prevalent in patients with severe asthma and is increasingly recognized as a potentially prevalent component of asthma physiology across severity stages. As providers progress through Global Initiative for Asthma (GINA) step therapy, eventually adding a long-acting muscarinic antagonist (LAMA) to an inhaled corticosteroid (ICS) and a long-acting β2-agonist (LABA), consideration must be given to each patient’s likely adherence to either closed or open therapy. In addition, underlying conditions, including SAD, may impact incidence of exacerbations despite adherence. If SAD is suspected, particle size may be an important factor in control, as smaller particles have been shown to achieve greater total lung deposition, farther distal airways penetration, and more peripheral lung deposition.
In this Q&A, John J. Oppenheimer, MD, defines SAD and addresses the question of how to best manage it as a treatable trait associated with well-controlled asthma. Corinne Young, MSN, FNP-C, FCCP, provides best practices for determining and improving patient adherence to therapy.
John J. Oppenheimer, MD, is an associate professor at the University of Medicine and Dentistry of New Jersey. He previously served as Director of Clinical Research at Pulmonary and Allergy Associates. Dr. Oppenheimer has coauthored more than 250 publications and is the Executive Editor of the Annals of Allergy Asthma and Immunology.

Q:
Why is SAD difficult to separate from asthma?
Dr. Oppenheimer: The real issue is that small airways—defined as beginning at the bronchioles and leading to the alveoli—have a diameter of less than 2 mm and can be major sites of inflammation and airflow resistance. They are known as the “silent zone” because SAD can progress without obvious symptoms. It’s hard to realize what’s going on in the small airways unless we do more sophisticated studies, so we typically rely on indicators or surrogates of small airways—such as forced expiratory flow 25% to 75%—which aren’t as accurate. Spirometry doesn’t really address the small, peripheral airways, so we either have to use more specialized tests such as oscillometry, which bounces low-frequency sound to measure resistance and reactance, or very expensive studies with hyperpolarized MRIs, using either helium or xenon, to determine molecule penetration in the different regions of the lung. We’ve learned over time that even those patients who we think are doing well can have peripheral airways disease.
An article by Aggarwal and colleagues noted that healthy individuals with normal lung functions may still have exercise-induced complaints, but accurate diagnosis is not possible until specialized testing focused on the peripheral airways is employed.1 It’s a lesson in humility—we’re really missing the root cause because we aren’t able to successfully interpret patient complaints. Even when you feel you have the answer, you may not be as correct as you think you are.
Q:
How is SAD related to asthma control or quality of life?
Dr. Oppenheimer: When we think about the stratification of disease, we’re really going from intermittent disease to severe disease. This is predicated on several factors: objective measures like lung function, frequency of exacerbations, and patient-reported outcomes using tools such as the Asthma Control Test or Asthma Control Questionnaire. These tests can be used to help stratify anywhere from the most mild to the most severe disease. Control is a really big issue, however, because we’re learning that sometimes the Asthma Control Test or tests like this may be diminished because of comorbidities or treatable traits such as reflux, sinus disease, or obesity. All of these things can bring about symptoms that may masquerade as asthma and/or exacerbate underlying asthma and that require treatment in order for the asthma to become controlled. So the question is whether we should be thinking about SAD as another treatable trait that is in the way of well-controlled asthma.
Q:
In the absence of symptoms, what is the relationship between SAD or lung function and exacerbation?
Dr. Oppenheimer: There are lots of moving parts here. We would like to believe that there’s a direct correlation between symptoms and lung function and exacerbation, but there may not be. One reason for this may be perception of dyspnea. In 2002 Magadle and colleagues reported on the perception of dyspnea in patients who had a near-fatal exacerbation.2 To do so, they had participants breathe against progressive resistance and rated the sensation of difficulty. Most patients had normal perception, but approximately 14% of patients had high perception, with breathing difficult with just minimal resistance, and approximately 26% had low perception, with reduction in acknowledgment of increased work to breathe. When following the group of poor perceivers, researchers found that this group was much more likely to require ICU admission as well as to die from asthma.
Q:
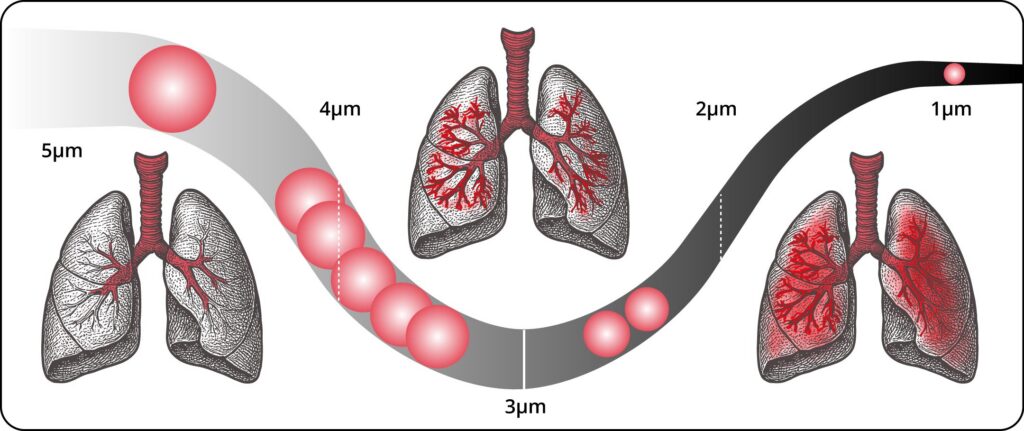
Where do smaller particles end up in comparison with the larger particles, and why is this important?
Dr. Oppenheimer: The respirable range for particle size is usually less than 5 µm to 0.5 µm. With larger-particle inhalers, the majority of the medication is never going to get to the lungs because it is captured in the mouth. As particles get smaller, there’s greater deposition in the peripheral airways until particles get so small, they don’t actually settle, which is known as Brownian Motion. The smaller and less dense a particle is, the better its ability to reach and settle in the small airways.3 In addition, smaller-particle formulations can often achieve effective asthma control at lower doses compared with larger-particle formulations, which may result in fewer adverse events, such as oropharyngeal candidiasis.4
Q:
How does the design of the single inhaler, or closed, triple therapy device matter to more efficient particle delivery?
Dr. Oppenheimer: It’s not just particle delivery; it’s also the ease of use. There are several closed triple therapies—an ICS, LAMA, and LABA all in one device—that are approved or in trials. I think it is important for patients to have a single inhaler with all the components because having separate inhalers is less efficient. People are not as adherent—there’s a plethora of data about that, but one study found that single-inhaler users were 31% more likely to be adherent than multiple-inhaler users.5 The CAPTAIN study compared triple therapy to dual therapy and found that the triple therapy improved lung function, symptoms, and asthma control. Patients on triple therapy also showed numerical improvements in annualized rates of moderate and/or severe exacerbations. It should be noted that the trial did not enrich for exacerbation so change was not as easy to see as in other studies.6
Recently, Mozaffaripour et al. measured small airway function in patients with asthma defined as GINA Steps 4 or 5 and that was poorly controlled on dual therapy.7 Patients began closed triple therapy and then underwent 129Xe MRI, computed tomography, and airwave oscillometry at different time points. Researchers found improved ventilation defect percentage and FEV1 at six weeks that lasted through the end of the study at one year. The airway markers—mucus score, lumen area, and total airway count—also showed significant improvement.
Even with these and other similar data, the problem is that we haven’t really shown the science yet regarding efficacy of the small-particle inhalers. The assumption that they work better is theoretical and based on the concern that larger particles don’t wind up where we want them in the lungs. These medicines are locally bronchodilating, so you need them to be at the site to work. If you can’t inspire or have respiration to that area, there’s no way in the world you’re ever going to get it to work. I think it makes a lot of sense, however, to want to use optimally sized particles knowing that inhalers themselves are inefficient. Any way I can improve efficiency at the end organ is a good day, but clinical validation is needed.
Q:
What are some best practices for therapeutic decision making for patients who have asthma/SAD and still experience exacerbations?
Dr. Oppenheimer: We know that there are many variables involved in the rate and severity of exacerbations, and they can be different for each patient. There’s poor adherence, which could be intentional or unintentional—just simple misuse of the device. Other confounders are cost of care and payer mandates regarding therapeutic switches, which also decrease adherence. So determining if the exacerbations are tied to adherence is important.
Determining whether the correct underlying cause or disease has been identified also is key. One study found that 33.1% of patients who had been previously diagnosed with asthma by a physician did not actually have asthma.8 In addition, there may be confounding medical issues, as discussed previously, that bring about symptoms that are misinterpreted as asthma.
So once I tackle those first two points, then I want to make sure that the medication is optimized with regard to delivery. That’s where the small particles come into play. Part of our job as clinicians is to find the right inhaler for the person, but we also have to optimize the device for the person. No one medicine works for everyone, and that’s why I’m employed. But through shared decision-making, we can try to find the right solution, validated by a decrease in nonadherence and an increase in patient satisfaction.
Corinne Young, MSN, FNP-C, FCCP, is the Founder and President of the Association of Pulmonary Advanced Practice Providers and a nurse practitioner at Colorado Springs Pulmonary Consultants. She has been a subinvestigator on numerous clinical trials and is a Fellow of the American College of Chest Physicians.

Q:
What are some best practices for educating patients with asthma/SAD about the role of particle size and lung deposition?
Young: I think it starts with explaining that not all inhalers are the same. Patients still have a lot of confusion, and rightfully so, about their inhalers because there are so many different types of inhalers on the market. Patients also often see multiple medical providers who prescribe different inhalers at different times. It’s not uncommon for a patient to come to me after being prescribed two or three different brands, using different delivery systems, within the same class of medication. I think the best practice is to first make sure your patients are educated on the different classes of therapy and then explain the different types and styles of inhalers and how to use those properly; because particle size or lung deposition are irrelevant if patients are not using the devices appropriately and correctly. After patients understand those issues, they can better understand why we might be escalating or de-escalating their therapy based on particle size and lung deposition.
Q:
How does the design of the closed triple inhaled therapy device matter to more efficient particle delivery?
Young: As we know, there are two triple therapies on the market currently, each in a different device: one is a dry powder inhaler (DPI) and the other is a metered dose inhaler (MDI). Over the years prior to triple therapy, we saw some patients struggle with DPIs because of the granular effect in their mouths or the taste associated with a DPI. Some patients are very sensitive to it and have increased coughing with it. Other patients may struggle with MDIs if spacers are not used or available. Some patients with arthritic hands may struggle actuating the device. So it’s really nice to have triple therapy options for all of these patients.
Q:
What are some best practices for teaching patients correct techniques for device use?
Young: Best practices for teaching device use would definitely include a live demonstration of how the device should be used, with the patient using the device in front of you to confirm their technique. Unfortunately, that approach is not always applicable or realistic, so it is also fine to walk through the steps without an actual device. Dr. Sandra G. Adams calls this “air guitar” because you don’t need to have a guitar to play the air guitar; same thing with an inhaler. The patient can still demonstrate their technique, including insertion of a spacer and shaking of the device (if applicable), inhalation, and holding of breath. Especially with an MDI and a spacer, we want to note how quickly the patient is inhaling because we want them to inhale very slowly if they are using a spacer.
There is a lot that can go wrong in the process of taking those medications, which is why we have a whole course during the CHEST Annual Meeting on inhaler technique. So many physicians show up and are blown away—they know all these inhalers are out there and they know how to dose them and prescribe them, but they’ve never physically used one or seen one before. It’s so important to get your hands on the device yourself so that you can educate the patient.
Q:
Outside of improved device techniques, what are some best practices for improving patient adherence?
Young: Inhaler education during the patient visit is reimbursable, but I think a lot of practitioners are not utilizing that code. However, inhaler technique education is worth it for your patient and because you do get reimbursed, it should always be included.
Oftentimes we question if a patient is getting benefit from a therapy, but is it not the right therapy for the patient or is it simply that the patient is not using the therapy appropriately? If we were to put patients on a blood thinner and it were as complicated as an inhaler, I don’t think we would question spending time to make sure the patient is using the correct technique. I do, unfortunately, think this type of patient-facing education is definitely lacking regarding inhalers.
It’s also important that patients understand the reason a certain therapy was selected for them. When we talk about triple therapy, we really are talking about daily maintenance therapy, so it is important that patients use these therapies consistently in order for them to see benefit.
Q:
What are some tips for providers regarding where to start and how to escalate therapies/devices?
Young: GINA step therapy can be a little much to take in initially. It’s not as straightforward as what we have in COPD because there are multiple tracks to pick from depending on your patient. These multiple tracks really enable individual tailoring of therapy, so you can find what will work best for the patient and within the constraints of insurance plans. Just because the GINA guidelines are there does not mean that insurance companies follow them, unfortunately. It can be a struggle to do what is recommended and what is best for the patient and what the insurance is going to allow you to do.
GINA Step 5 is for difficult-to-treat or severe asthma, which applies to those patients for whom we recommend triple therapy. This is after patients have worked their way through medium- or high-dose ICS therapy plus LABA therapy, but their asthma is still not in good control. At that point, we step up to adding on the LAMA therapy, and we may consider additional testing for alternative or additional diagnoses, such as SAD.9
Q:
What else do you think about when a patient still has exacerbations despite adherence?
Young: First we want to check technique. If they’re spraying it in their ear, it doesn’t matter how compliant they are—it’s not going to do very much for them. Then we want to see what else is influencing their asthma. We know a lot of asthma is all about the trigger, so we must investigate if something has changed with their environment or their exposures that is causing their asthma to be more active. From there, we would definitely look at biomarkers if that has not already been done. We want to establish if they have type 2 asthma and if their absolute eosinophil counts are elevated. Then we can explore other comorbid conditions that can really influence their asthma, such as reflux, that has not been addressed. There might be silent aspiration issues or upper airway dysfunction, like vocal cord dysfunction, that can mimic asthma symptoms. Life is not fair, and patients can have multiple conditions simultaneously that may, unfortunately, influence each other. Lastly, I would also look at their cardiac function.
References
1. Aggarwal B, Mulgirigama A, Berend N. Exercise-induced bronchoconstriction: prevalence, pathophysiology, patient impact, diagnosis and management. NPJ Prim Care Respir Med. 2018;28:31. doi:10.1038/s41533-018-0098-2
2. Magadle R, Berar-Yanay N, Weiner P. The risk of hospitalization and near-fatal and fatal asthma in relation to the perception of dyspnea. Chest. 2022;121(2):329-333. doi:10.1378/chest.121.2.329
3. Spangler DL. The role of inhaled corticosteroids in asthma treatment: A health economic perspective. Am J Manag Care. 2012;18(2Suppl):S35-S39.
4. Gentile DA, Skoner DP. New asthma drugs: small molecule inhaled corticosteroids. Curr Opin Pharmacol. 2010;10(3):260-265. doi:10.1016/j.coph.2010.06.001
5. Busse WW, Abbott CB, Germain G, et al. Adherence and persistence to single-inhaler versus multiple-inhaler triple therapy for asthma management. J Allergy Clin Immunol Pract. 2022;10(11):2914-2915. doi:10.1016/j.jaip.2022.06.010
6. Oppenheimer J, Kerstjens HA, Boulet L-P, et al. Characterization of moderate and severe asthma exacerbations in the CAPTAIN study. J Allergy Clin Immunol Pract. 2024;12(9):2372-2380.e5. doi:10.1016/j.jaip.2024.05.019.
7. Mozaffaripour A, Tcherner S, Duron E, et al. Airway mucus and 129Xe MRI ventilation after single inhaler triple therapy in asthma. ERJ Open Research. 2025;11(5):01333-2024. doi:10.1183/23120541.01333-2024
8. Aaron SD, Vandemheen KL, FitzGerald JM, et al. Reevaluation of diagnosis in adults with physician–diagnosed asthma. JAMA. 2017;317(3):269-279. doi:10.1001/jama.2016.19627
9. Global Initiative for Asthma. Difficult-to-treat & severe asthma in adolescent and adult patients, diagnosis and management: a GINA pocket guide for health professionals. Version 2.0. Published April 2019. https://ginasthma.org/wp-content/uploads/2019/04/GINA-Severe-asthma-Pocket-Guide-v2.0-wms-1.pdf
10. Henao MP, Kraschnewski JL, Bolton MD, Ishmael F, Craig T. Effects of inhaled corticosteroids and particle size on risk of obstructive sleep apnea: a large retrospective cohort study. Int J Environ Res Public Health. 2020;17(19):7287. doi:10.3390/ijerph17197287
